Breathing apparatus. Cylinders, gases and decompression sickness
Dedicated to everyone who has mastered the second part of the cycle on breathing apparatus.
In the second part, I talked about the device of open-circuit breathing apparatus (OTs). But I did not pay attention to the cylinders for these devices. But the birth of autonomous OC apparatuses led precisely to the appearance of high-pressure cylinders.
I will not go into history, I will describe only the main cylinders currently used in DA.
1. Steel cylinders. Working pressure 200-300 atm. Made of alloy steel. They always have negative buoyancy in water. They rust, therefore, they are demanding on the integrity of the outer paintwork and on the absence of water in the clogged air. Steel cylinders have the longest service life. Until recently, plastic shoes were used in steel cylinders due to the bottom rounding so that the cylinder could stand vertically on the bottom. Now there are flat-bottomed cylinders.
2. Aluminum cylinders. Working pressure ~ 210 atm. They have the largest weight per unit volume in air. The service life is slightly less than that of steel. In water, in an empty state, they have positive (!) Buoyancy. Yes Yes. The empty bottle floats up. Even in fresh water. The bottom is usually flat. They rust badly. For this they are very fond of diving centers and dive boats. They do not require as much attention to themselves as steel ones.
3. Composite (metal-composite) cylinders. The composite in them is something like fiberglass in several layers, filled with epoxy. Working pressure 200-300 atm. They have the lowest weight per unit volume. Fully composite does not rust. At all. Metal-composites have a thin-walled steel bulb inside, and a composite outside. When water gets inside, steel is corroded accordingly. They have the shortest service life (5 years in the Russian Federation). They are not subject to pressure testing (hydrotest). These cylinders are mainly used for work in the air.
In general, now at least the Ministry of Emergency Situations has completely abandoned closed-circuit devices. Although the purchase of ECCR rebreathers is planned for deep-sea work. Moreover, it is made in Russia. In diving, such composites are rarely used due to their high buoyancy. They have a big plus: when the balloon explodes, they do not give fragments. But they are afraid of mechanical damage.
Yes, a few words about hydrotest or, as it is called, pressure testing.
The valve is unscrewed from the balloon, a fitting is screwed in its place, the balloon is filled with liquid and placed in a bath of water. Then a pressure is applied to the cylinder, usually 1,5 times the operating pressure. That is, 300 atm is injected into a cylinder with a working pressure of 450 atm. In this case, the change in volume is measured (by the amount of liquid that has filled the cylinder with increasing pressure).
There are norms for the increase in volume. If the norm is exceeded or destruction occurs, the cylinder is discarded.
The liquid is used because of its practical incompressibility. After all, if the cylinder collapses with the gas inside, then there will be a big boom. And with liquid, it will just crack. By the way, household gas cylinders (which are propane-butane) must also undergo a hydrotest, but they usually “hammer” everything into it.
By the way, perhaps someone will be interested in the question why, for example, 400-atmospheric cylinders are not used, although they really exist?
Firstly, a 400 atm compressor is rather complicated, expensive and bulky.
Secondly, one should not forget about the van der Waals forces. This is when a gas under high pressure begins to acquire the properties of a liquid. And if at 200 atm these forces are practically invisible, then, for example, at 300 atm it will already be about 10%, and at 400 - already about 18%.
That is, having a 10 liter bottle filled up to 200 atm, we have 2 liters of gas, at 000 atm - 300 2 liters, and at 810 atm in the same bottle there will be 400 3 liters. Well, all the high-pressure fittings on the gearboxes will have to be decently redone.
That's it, finished with the OTs.
But before moving on to the closed cycle, I will allow myself to digress a little and recall the reasons for using and improving these rather complicated and expensive devices.
CST
As I already said in the first part of the review, for a person to breathe, it is necessary to ensure that a certain amount of gas circulates through the lungs. Gas to the lungs must be supplied at a pressure equal to the ambient pressure plus 1 atmosphere. And if the diver has plunged, say, 30 meters under the water, then gas will be supplied to his lungs by the OTs breathing apparatus under a pressure of 4 ata (do not forget about +1 ata on the surface, yeah).
What will happen to the diver's body when breathing gas with increased pressure?
And there will be an oversaturation of the body with gas. After all, it is no secret to anyone that the human body consists mainly of water, and the saturation of a liquid with a gas is directly proportional to the pressure at which this saturation occurs.
Of course, the saturation process does not happen quickly. The excess pressure of the inhaled gas is transmitted through the alveoli to the blood. The oversaturated blood, circulating through the body, saturates other tissues. Moreover, studies have shown that different tissues are saturated in different ways.
Rapidly saturated tissues: blood, skin, adipose tissue. Slow tissues: bone, connective. Modern decompression algorithms use up to 16 types of fabrics. The process of increasing external pressure, compression, can occur quite quickly. This does not cause any unpleasant consequences for the body.
But the reverse process, decompression, can no longer occur as quickly as compression.
This is where the comparison of the decompression process with a bottle of soda comes in. By drastically reducing the external pressure, we provoke the release of the dissolved gas from the liquid. In the form of bubbles.
Probably, no one needs to explain what is fraught with the formation of gas bubbles in the bloodstream?
Blood clots. Thrombi clog up blood vessels, at first small, then ever larger, until they reach the vessels leading to / from the heart. Death is coming.
There are less fatal consequences of DCS. For example, deformation of various tissues, say, cartilaginous, due to the formation of bubbles in these tissues. And the most unpleasant symptom of DCS is pain caused by the pressure of gas bubbles formed in the tissues on the nerve endings. Moreover, these painful sensations are well, very strong.
To avoid the occurrence of DCS, decompression tables were developed, and later, with the advent of computers, decompression algorithms. These tables indicated the depth (pressure) and the time at which the diver had to stop there before going to the surface.
In the same tables, the gas mixture was taken into account. Indeed, if a mixture with an increased oxygen content, up to 100%, is used for performing decompression procedures, the decompression time is reduced. And this happens because oxygen, unlike inert gases, is partially absorbed by the body for its metabolic processes.
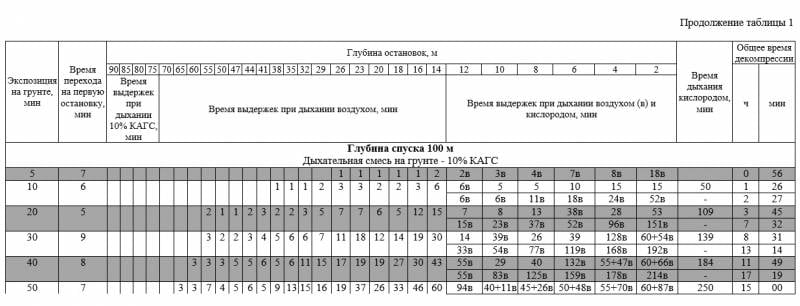
This is a fragment of decompression tables from the Navy's PVS in the 2002 edition. Pay attention to the second line (10 minutes bottom time). When using air as a decompression gas, the decompression time is 2 hours 27 minutes, in the case of using oxygen, this time will be 1 hour 26 minutes! Here you can also note the depth of the beginning of oxygen breathing - it is 10 meters. That is, the partial pressure of oxygen at this depth will be 2 ata, which is dangerous according to the norms of amateur diving.
It should also be noted that there are no decompression limits.
These are such values of pressure / time, while being in which a diver can quickly (without observing decompression procedures) switch to breathing at a pressure of 1 ata without consequences for the body.
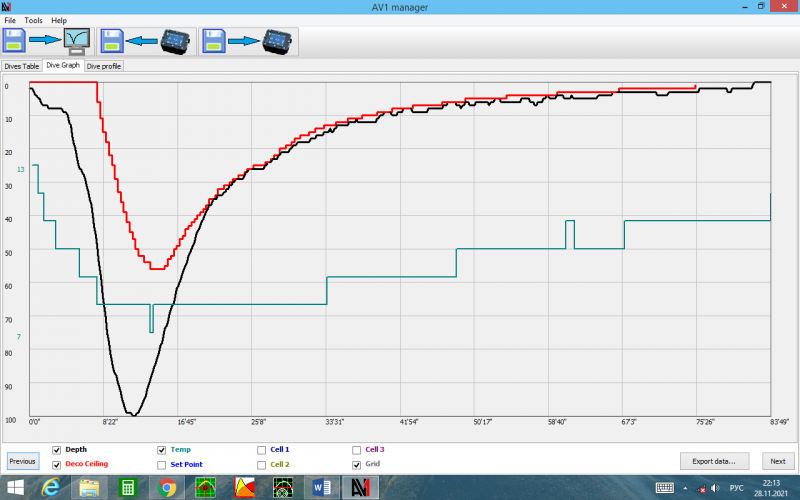
This is a profile of a real dive to a depth of 100 m in an alpine environment (800 m above sea level). The black curve is the depth, the red curve is the deco obligations, the green is the water temperature. Gases: KAGS 13/63; CAS 41; oxygen. The no-decompression limit is clearly visible, this is the zero segment of the red line
Toxic effect of gases at elevated pressure
It would seem that everything is simple: we breathe pure oxygen and get the minimum decompression time.
But, it turns out, not everything is as good as it might seem.
At the dawn of the development of diving, in experiments with breathing oxygen under excessive pressure, the testers noted unpleasant changes in the condition of the subjects. So, when some pressure values were reached, the divers began to have muscle cramps up to a complete loss of control over motor functions. The negative effect of oxygen on the efficiency of gas exchange in the lungs was also noted. That is, with prolonged exposure to an increased partial pressure of oxygen on the alveoli, they partially lost their gas exchange capabilities.
A safe value of the partial pressure of oxygen in the respiratory mixture was established experimentally. So, for a long exposure, this value is 1,4 ata, for a short one - 1,6 ata.
It also turned out that under excess pressure, an inert gas nitrogen causes the so-called. "Nitrogen narcosis", similar in symptomatology to the effects of alcohol. Inhibition of reaction, inadequate assessment of the situation.
Probably not worth telling how this can turn out for a diver?
A nitrogen partial pressure of ~ 3 ata is considered safe.
At the same time, I want to note that the values of the limiting pressures of both oxygen and nitrogen can vary depending on the organizations that regulate them.
For example, in the Russian Armed Forces, a value of 3 ata is considered permissible for breathing with 100 percent oxygen. Such a high PPO2 value is not found anywhere else in the world, apparently, our divers have some kind of special organism.
So, to reduce the content of oxygen and nitrogen in the breathing mixture, they began to use helium. Helium practically lacks both the effect of anesthesia and the toxic effect on the body. Respiratory mixtures of nitrogen, helium and oxygen are called KAGS (trimixes), from oxygen and helium - KGS (helioxes).
When calculating the composition of the breathing mixture, the percentage of oxygen and nitrogen in it is usually made the maximum allowable, based on the diving conditions, of course. Oxygen is used to shorten the decompression time, and nitrogen is used because helium is expensive.
In Russia, helium is relatively cheap because of the way it is mined. But abroad the price was around 7 cents per liter (four years ago, Egypt). That is, the cost of gas in a 2x15x200 pair with a mixture of 12% O2, 70% He and 18% N2 would be about $ 300.
Moreover, most of this gas will simply be thrown out, because during breathing, a person pumps 10-50 liters of gas through the lungs. Why is there such a difference, you ask. And it's all about the physiology and state of the body. The greater the volume of the lungs and muscle mass, the greater the consumption will be. For example, graceful girls, small lung volume and small muscle mass, gas consumption is usually very low, in physically developed men with large lungs, on the contrary. Well, gas consumption at rest can be 5-6 times less than in a state of stress. Physical work also leads to an increased consumption of breathing gas, but not as much as during stress.
It is precisely the issues of saving expensive breathing mixtures that have led to the emergence (or development) of apparatus for a closed breathing cycle - rebreathers. Although, strictly speaking, rebreathers were the first self-contained breathing apparatus.
And I will talk about the design and types of rebreathers in the next part.

Information