Side effect of sedative. The Contergan disaster
Teratogen number 1
The first wake-up call regarding thalidomide was back in the 1956 year, before it was launched into over-the-counter distribution. One Chemie Grunenthal employee decided that his pregnant wife needed to treat morning sickness and malaise with the new Contergan (the trade name for the first version of thalidomide). The daughter was born without ears!
Then, of course, no one revealed a causal relationship, and a year later the drug went into series. It is noteworthy that initially the drug was considered as an anticonvulsant, but tests showed not the highest efficacy of thalidomide in this direction. Therefore, it was decided to use its "side" property to soothe patients and give deep sleep. In the pharmaceutical market of that time, Contergan was almost the most effective drug, causing rave reviews from both patients and their doctors. Pregnant women have successfully used the novelty in the fight against morning sickness, insomnia and anxiety.

Contergan Sedative Pack
It is worth mentioning that no one conducted preliminary tests of the drug on pregnant animals, and even less so on women “in position”. And thalidomide conquered new markets every year: at the peak of its career, it was sold in more than forty countries of the world. Except the USA. But more on that later. In particular, only in the UK could thalidomide be found on drugstore shelves under the trade names Distaval (Forte), Maval, Tensival, Valgis or Valgraine. Four years after the launch of thalidomide drugs on the market, a German doctor, Hans-Rudolf Wiedemann, indicated an abnormally high percentage of congenital malformations and directly associated this phenomenon with a side effect of a sedative. Prior to this, many doctors from Germany pointed out the increasing incidence of stillbirth and deformity, but attributed this to atmospheric nuclear tests in the United States. In 1958, they even sent requests to the defense department.
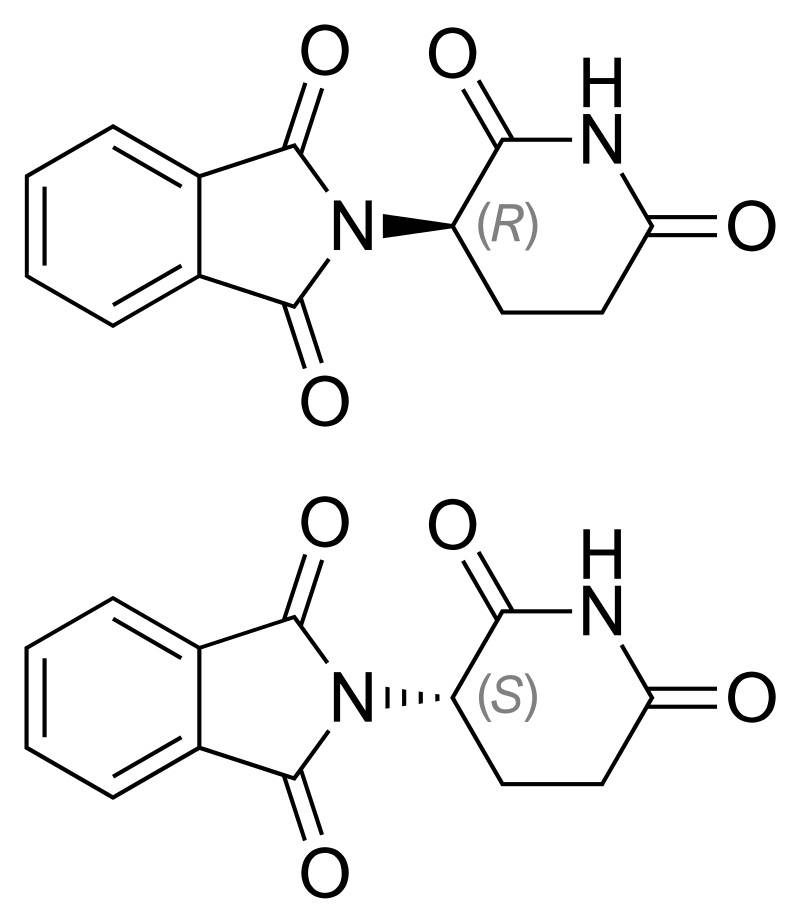
Structural formulas of thalidamide molecules. One of them has a medicinal effect, the second - teratogenic

Chemie Grunenthal Logo
The action of the teratogen was terrible: the fetus inside the mother lost its eyes, ears, internal organs and often came to light already dead. The most common is phocomelia, or the syndrome of seal extremities, when the newborn was either completely deprived of the limbs, or they were underdeveloped. At the same time, thalidomide did its dirty work not only in the female body, but also disrupted sperm formation, dooming future fathers to inferior offspring.
In this stories There is an interesting person - Australian gynecologist William McBride. In December 1961, he published an article in the reputable journal The Lancet on the teratogenic effect of the sedative Chemie Grunenthal. It was from him and from the mentioned Hans-Rudolph Wiedemann that the world community learned about the terrible drug. McBride immediately became famous and even received a prestigious French medal and cash prize from L'Institut de la Vie. But fame is very variable - after some time, the thalidomide scandal subsided, and they forgot about McBride.
The gynecologist later tried to draw attention to his person with the supposedly existing connection between the deformities and methods of certain antidepressants, but nothing was proved. And in 1981, he suddenly accused the drug Debendox of a similar teratogenic effect on thalidomide, fabricated test trials and published all this. Only in 1993, doctors and pharmacists understood fraud and deprived the former celebrity of the right to practice medicine until 1998.
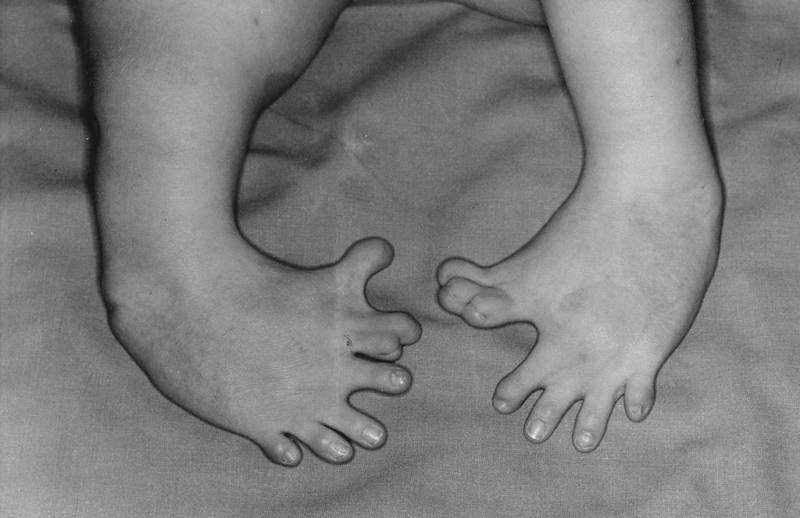
Typical fetal developmental disorders

Orthopedic costume for thalidomide victims

German director Niko von Glazov
But back to thalidomide. He was removed from the market in December 1961, immediately after the publication of the publication in the reputable medical journal The Lancet, but the picture of his atrocities was amazing. About 40 thousand people were affected by peripheral neuritis, the most harmless side effect of thalidomide. More than 10 thousand children were born (data differ in sources) with severe developmental disabilities, of which more than half survived. Now many of them have been able to sue Chemie Grunenthal for compensation and life-long maintenance. The German government also supports birth-disabled people with monthly allowances, some of which are barely enough. So, in 2008, several victims of thalidomide immediately demanded three times higher disability pensions and went on an unlimited hunger strike.
Francis Kesley - US Savior
Why is thalidomide such a powerful teratogen? The mechanism of its action was discovered just nine years ago, and before that they only knew that a molecule of a substance can exist in two optical isomers (this is the course of the school chemistry program). One form heals, and the other, accordingly, cripples. Moreover, even a simple purification of the drug from teratogenic isomers will not help: our body will independently produce a particularly dangerous molecule from its useful form. After revealing publications about the Contergan disaster, many medical centers began testing thalidomide-based drugs in pregnant rodents. And it turned out that mice do not have any teratogenic effect even at exorbitant dosages. That is, even if Chemie Grunenthal had conducted Contergan preliminary tests on laboratory animals, a dangerous medicine would have successfully passed them. Even repeated studies on pregnant monkeys did not reveal any contraindications for the withdrawal of the drug to world markets.
However, thalidomide still could not convince one pharmacist of its own safety. Francis Kesley, an employee of the US Food and Drug Administration (FDA), had expressed serious doubts about the safety of the drug for pregnant women even before the start of the Contergan scandal. This was indicated by mild side effects or it was Francis's professional instinct, we cannot say for sure, but the drug was not allowed on the US market. A small number of free lots for testing does not count. And when the whole world found out about the disaster with thalidomide, Kesley became the country's national hero. It turned out that the researcher made her decision under pressure from Richardson-Merrell (a marketing division of Chemie Grunenthal), which in every way imposes a new drug on the FDA. If Kesley hadn’t sent the drugs for an additional study in 1960 (which, as you know, would have led nowhere), time would have been lost and thalidomide would have ended up in pharmacies. But while the test cycle on pregnant animals was started, while the results were being evaluated, December 1961 of the year came, and all further work turned out to be superfluous. John Kennedy personally presented Francis Kesley with a state award for professionalism that has saved thousands of American lives.
A lawsuit was launched against Chemie Grunenthal, but the real culprits were not identified. Rumor had it that employees had destroyed a lot of documentation on the test results of the drug on time. Be that as it may, the company paid 100 million marks to the thalidomide victim fund, which still pays lifelong pensions to people with disabilities around the world.
The Contergan catastrophe forced tightening of control over medicines and sharply increased the costs of pharmaceutical companies for the development of new drugs. The most interesting thing is that doctors around the world still prescribe drugs for thalidomide to their patients. Of course, not to expectant mothers and not as sleeping pills, but as a powerful anti-cancer agent. There is research that the notorious thalidomide can almost be cured of AIDS.





Information