Rocket Fuel Saga
"... And there is nothing new under the sun"(Eccliaste 1: 9).
About fuels, missiles, rocket engines was written, written and will write.
One of the first works on fuel rocket engines can be considered the book of V.P. Glushko "Liquid jet fuel", published in 1936

For me, the topic seemed interesting, related to my former specialty and study at the university, much less the “trailing” of my younger offspring: “Chef, let's knead it and run the thread, but if it's too lazy, then ourselves let's figure it out. Apparently Lavra Extremes from "Lin Industrial" do not give rest.
"Consider" will be together under strict parental control. Hands feet must be intact, strangers all the more.
"The key to start" ... "Go"! (Yu.A. Gagarin & S.P. Korolev)
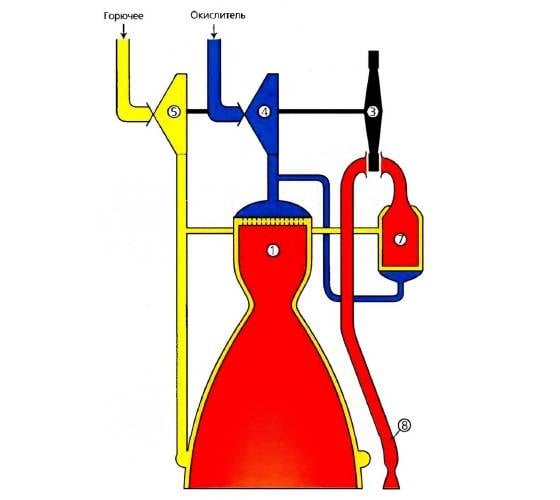
Whatever type of RD (scheme, nature of the process) is not used in rocket technology, its intended purpose: the creation of thrust (force) by converting the initial energy stored in the RT to the kinetic energy (Ec) of the jet stream of the working fluid.
Ek jet of the jet in the RD will convert different types of energy (chemical, nuclear, electric).
For chemical engines, fuel can be divided by phase state: gaseous, liquid, solid, mixed.
Part №1 - fuel for rocket engine or liquid rocket fuel
Classification of chemical fuels for rocket engines (common):
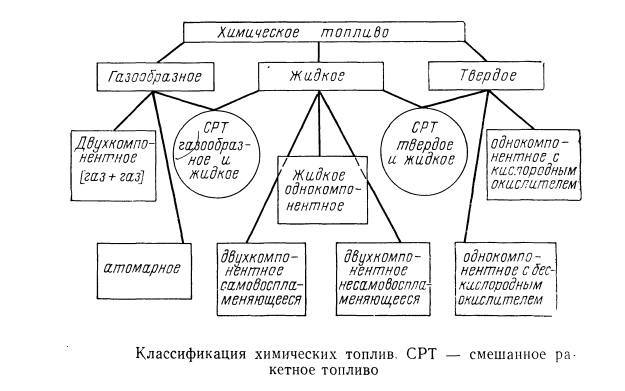
->Terms and abbreviations.
Additionally (HTML tags on TopWar of the wrong system, therefore spoilers and katas have to be organized like this):
Specific impulse (Iud).
Jet thrust (P or Fp).
The stoichiometric ratio of fuel components (Km0)(more-click-the ratio of the mass of the oxidizer to the mass of fuel during stoichiometric reactions.
The composition of the fuel is combustible and non-combustible parts (in general).
Types of fuels(in general).
The chemical source of thermal energy for RD in general can be considered the chemical reaction of the components of the RT.
I'll start broadcasting with Km0. This is a very important correlation for RD: fuel can burn differently in RD (a chemical reaction in RD is not normal firewood burning in the fireplacewhere air is used as an oxidizing agent). Combustion (more precisely, oxidation) of fuel in the chamber of a rocket engine is, first of all, a chemical oxidation reaction with the release of heat. And the course of chemical reactions essentially depends on how many substances (their ratio) reacts.
How to fall asleep on the protection of the course project, exam or test. / Dmitry Zavistovsky
The value of Km0 depends on the valence that chemical elements may exhibit in the theoretical form of a chemical reaction equation. Example for ЖРТ: АТ + НДМГ.
An important parameter is the coefficient of excess oxidant (symbolized Greek "α" with the index "approx.") And the mass ratio of the components of the Km.
Km = (dm. / Dt) / (dmg ../ dt), i.e. the ratio of the mass flow rate of the oxidizer to the mass flow of fuel. It is specific to each fuel. In the ideal case, is the stoichiometric ratio of oxidant and fuel, i.e. indicates how much kg of oxidizer is needed to oxidize 1 kg of fuel. However, the real values are different from the ideal. The ratio of real Km to the ideal is the coefficient of excess oxidant.
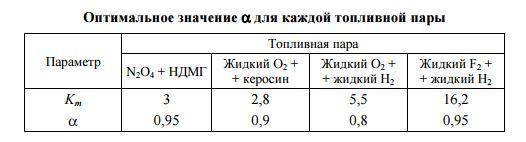
As a rule, αapp. <= 1. And that's why. The dependences Tk (αok.) And Isp. (Αok.) Are nonlinear and for many fuels the latter has a maximum at αok. not with stoichiometric mixing ratio, i.e. max. values Iud. are obtained with a slight decrease in the amount of oxidizing agent in relation to the stoichiometric one. A little more patience, because I can not get around the concept: enthalpy. It is useful in the article, and in everyday life.
Briefly, enthalpy is energy. For the article are important her two "incarnation":
Thermodynamic enthalpy- the amount of energy expended on the formation of a substance from the original chemical elements. For substances consisting of identical molecules (H2, O2 and so forth), it is zero.
Enthalpy of combustion- makes sense only under the condition of a chemical reaction. In reference books one can find the values experimentally obtained under normal conditions. Most often for combustible it is full oxidation in the environment of oxygen, for oxidizing agents - oxidation of hydrogen by a given oxidizing agent. Moreover, the values can be both positive and negative, depending on the type of reaction.
"The sum of the thermodynamic enthalpy and enthalpy of combustion is called the total enthalpy of the substance. Actually, this value is used in the thermal calculation of the LRE chambers."
-as a power source;
-as to the substance that has to be (at this level of technology development) used to cool RD and THA, sometimes to pressurize tanks with RT, to provide it with volume (RN tanks), etc .;
-as to a substance outside the LRE, i.e. during storage, transportation, refueling, testing, environmental safety, etc.
Such gradation is relative conditional, but in principle reflects the essence. I will name these requirements as follows: №1, №2, №3. Someone can add to the list in the comments.
These requirements are a classic example. "Swan cancer and pike"that "pull" the creators of RD in different directions:
# From the point of view of the source of energy LRE (№1)

Those. need to get max. Id. I will not continue to hammer heads with everything, in general:

With other important parameters for №1 we are interested in R and T (with all indices).
Need to: the molecular mass of the combustion products was minimal, the maximum was the specific heat content.
# From the point of view of the PH designer (№2):
TCs should have a maximum density, especially in the first stages of rockets, since they are the most voluminous and have the most powerful taxiways, with a large second consumption. Obviously, this is inconsistent with the requirement under No. XXUMX.
# With operational tasks important (#3):
- chemical stability of TC;
-easy refueling, storage, transportation and manufacturing;
-ecological safety (in the whole "field" of application), namely toxicity, cost of production and transportation, etc. and safety when operating the taxiway (explosive).
Of course, this is only the tip of the iceberg. Additional requirements are also getting in here, because of which CONSENSUSES and COMPROMISES should be sought. One of the components must have satisfactory (better excellent) properties of the cooler, since at this level of technology, it is necessary to cool the CS and the nozzle, as well as to protect the critical section of the taxiway:
In the photo, the LRE XLR-99 nozzle: the characteristic feature of the American 50-60 LRE design is clearly visible - a tubular chamber:
It is also required (as a rule) to use one of the components as a working fluid for a THA turbine:
For fuel components, "saturated vapor pressure is of great importance (this is roughly the pressure at which a liquid starts to boil at a given temperature). This parameter greatly influences the design of pumps and the weight of tanks." / S.S. Fakas /
An important factor is the aggressiveness of the TC to materials (KM) LRE and tanks for their storage.
If TCs are very "harmful" (like some people), then engineers have to spend money on a number of special measures to protect their structures from fuel.
-self-igniting fuel components like two-faced Janus: sometimes necessary, and sometimes it hurts. There is another nasty property: explosiveness
For many industries using missiles (military or deep space)
the fuel is required to be chemically stable, and its storage, refueling (in general, everything that is called: logistics) and recycling do not cause a "headache" among operators and the environment.
An important parameter is the toxicity of combustion products. Now it is very relevant.

The cost of production of both the TC and the tanks and CM, which satisfy the properties (sometimes aggressive) of these components: the load on the economy of the country, claiming the role of "space cab".
There are many of these requirements and, as a rule, they are antagonistic to each other.
Conclusion: the fuel or its components must have (or possess):
2. The greatest density, minimal toxicity, stability and low cost (in production, logistics and disposal).
3. The highest value of the gas constant or the lowest molecular weight of the combustion products, which will give Vmax flow and excellent specific thrust impulse.
4. Moderate combustion temperature (no more than 4500K), otherwise everything will burn or burn. Do not be explosive. Self-ignite under certain conditions.
5. Maximum rate of combustion. This will provide the minimum weight and volume of the COP.
6. Minimum ignition delay period smooth and reliable launch of the taxiway plays a significant role.

A whole heap of problems and requirements: viscosity, melting and solidification, boiling temperature, evaporation, vapor pressure and latent heat of vaporization, etc. etc.
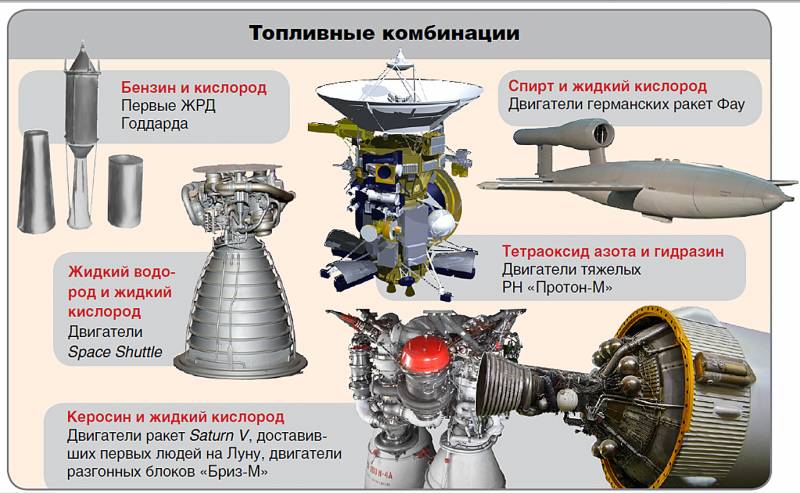
Compromises vividly manifest themselves in Iud .: TK high density (kerosene + LOX), as a rule, are used on the lower stages of the PH, although they lose to the same LH2 and LOX, which in turn are used in the upper stages of the PH ("Energy" 11-25).

And again, a beautiful couple LH2+ LOX can not be used for deep space or for long-term stay in orbit (Voyager-2, Briz-M upper stage, ISS, etc.)
Awesome moment of undocking the GOES-R meteorological satellite from the Centaur upper stage of the Atlas V 541 launch vehicle (GOES-R Spacecraft Separation)
LCT classification - most often by saturated vapor pressure or triple point temperatureand, more simply, boiling point at normal pressure.
High-boiling components ЖРТ.
Chemical substance having a maximum operating temperature at which saturated steam pressure (I will refer to Pnp) in the tanks of the rocket is significantly lower than the permissible level of pressure in the tanks because of their structural strength.
Example:
Accordingly, they are stored without any special manipulations with cooling tanks.

I personally like the term “container” more. Although it is not entirely correct, but it is close to the everyday value. This is the so-called. long-life TC.
Low boiling components ЖРТ.
Here, the RNP is close to the maximum allowable pressure in the tanks (by the criterion of their strength). Storage in sealed tanks without special measures for cooling (and / or cooling) and returning the condensate is impossible. The same requirements (and problems) with the LRE fittings and refueling / discharge pipelines.
Example:
The Ministry of Defense of the Russian Federation (MO of the Russian Federation) considers low-boiling components allwhose boiling point below 298K under standard conditions.
Cryogenic components ЖРТ.
In fact, it is a subclass of low-boiling components. Those. substances having a boiling point below 120K. The cryogenic components include liquefied gases: oxygen, hydrogen, fluorine, etc. To reduce evaporation losses and increase the density, it is possible to use a cryogenic component in the bulk state, as a mixture of the solid and liquid phases of this component.
Special measures are required during transportation, refueling (cooling down of tanks and highways, heat-insulation of LRE valves, etc.) and discharge.
The temperature of their critical point is much lower than operational. Storage in sealed tanks PH is impossible or very difficult. Typical representatives of oxygen and hydrogen in the liquid phase state.
Further I will use the American style of their designations LOX and LН2 respectively. Or so LCD and LW.
Our "handsome" RD-0120 (hydrogen-oxygen):
It can be seen that it is outside (valves, highways) completely filled with insulating material.
When the components of the RT are found in the COP LRE (intelligently react), they should be divided into:
STK: upon contact of the oxidizer and the fuel in the liquid state they ignite (in the whole range of operating pressures and temperatures).
This greatly simplifies the RD ignition system, however, if the components meet outside the combustion chamber (leaks, accidents), there will be a fire, or a big woman. Quenching difficult.

Example:N204 (nitrogen tetraxide) + MMG (monomethylhydrazine), N204 + N2H4 (hydrazine), N2О4+ UDMH and all fluorine based fuels.
OSTK: special measures must be taken to ignite. Non-flammable fuels require an ignition system.
Example:kerosene + LOX or LH2+ Lox.
NTK: I think there are no comments here. It requires either a catalyst, or a constant ignition (or temperature and / or pressure, etc.), or a third component.
Ideal for transportation, storage and leak-proof.
Another separation option is based on the level of energy characteristics of the LRT:
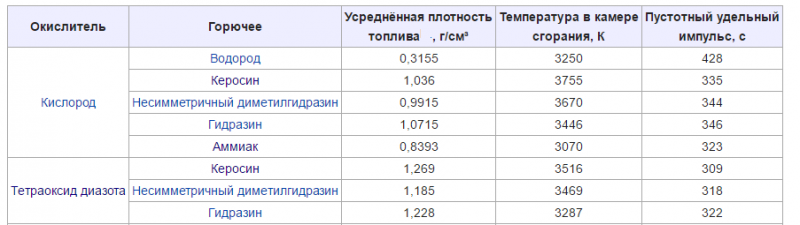
* average energy (with average specific impulse— (0Xnumx) + kerosene, N204 + MMG and others.);
* high energy (high specific impulse: (02) W + (N2) F, (F2) W + (N2) OK and others.).
On toxicity and corrosivity of components distinguish ЖРТ:
* on non-toxic and non-corrosive fuel components - (02g) hydrocarbon fuels, etc .;
* on toxic and corrosion-active components of the fuel - MMG, UDMH and especially (F2) g.
According to the number of fuel components used, one-, two-, and three-component control systems are distinguished.
In one-component remote control, which most often use pressure feed.
At the initial stage of development of auxiliary one-component remote control systems for satellites, spacecraft and spacecraft, high-concentration (80 ... 95%) hydrogen peroxide was used as a single-component fuel.
At present, such auxiliary propulsion systems are used only in the stage orientation systems of some Japanese PH.

For the remaining auxiliary one-component DUs, hydrogen peroxide is “displaced” by hydrazine, while an increase in the specific impulse by about 30% is ensured.
The most widely mankind uses two-component TCs, which possess higher energy characteristics as compared to single-component ones. But two-component fuel rocket engines are more complex in design than single-component ones. Due to the presence of oxidizer tanks and fuel, more complex piping system and the need to ensure the required ratio of fuel components (coefficient Kto). In the remote control of the satellite, KK and KA often use not one but several tanks of oxidizer and fuel, which further complicates the system of pipelines of two-component remote control.

Three component RT in development. This is a real exotic.
RF patent for a three-component rocket engine.
The scheme of this LRE .
Such LRE can be classified as multi-fuel.
LRE on a three-component fuel (fluorine + hydrogen + lithium) was developed in OKB-456.
Two-component fuels consist of an oxidizer and fuel.
LRE Bristol Siddeley BSSt.1 Stentor: two-component LRE (H2O2 + kerosene)
Oxidizing agents
Oxygen
Liquid, rather than gaseous oxygen-liquid oxygen (LOX-briefly and everything is clear) is used in the LRE.
Molecular mass (for the molecule) is 32g / mol. For lovers of accuracy: atomic mass (molar mass) = 15,99903;
Density = 1,141 g / cm³
Boiling point = 90,188K (−182,96 ° C)

In terms of chemistry, the ideal oxidant. It was used in the first ballistic missiles of the FAA, its American and Soviet copies. But his boiling point did not suit the military. The required operating temperature range is –55 ° C to + 55 ° C (long preparation time for launch, short time on alert).
Very low corrosivity. Production has long been mastered, the cost is small: less than $ 0,1 (in my opinion, cheaper than a liter of milk at times).
Disadvantages:
Cryogenic - necessary cooling and constant refueling to compensate for losses before the start. Also can spoil other TK (kerosene):

In the photo: shutters of kerosene refueling devices (ZU-2), for 2 minutes before the end of the cyclogram when performing the operation CLOSE due to icing not fully closed. At the same time, due to icing, the signal about the TUA exit from the launcher did not pass. Start held the next day.

The unit-tanker RB liquid oxygen removed from the wheels and installed on the foundation.
Difficult to use as a coolant and the COP nozzle rocket engine.
See
Now everyone is studying the possibility of using supercooled oxygen or oxygen in the slug-shaped state, in the form of a mixture of solid and liquid phases of this component. The view will be about the same as this beautiful ice slush in the cove to the right of Shamora:
Dream up: instead of H2About imagine LCD (LOX).
Scrooping will increase the overall density of the oxidizer.
Example of cooling down (hypothermia) BR P-9А: for the first time, it was decided to use supercooled liquid oxygen as an oxidizer in a rocket, which reduced the total preparation time of the rocket for launch and increased its combat readiness.

Note: for some reason, for the same procedure, he bent (almost "chmoril") Ilona Mask famous writer Dmitry Konanykhin.
Cm:
In defense of the pasta monster Ilona Mask put in a word. Part of 1
In defense of the pasta monster Ilona Mask put in a word. Part of 2
Ozone-O3

Fluid density at -188 ° C (85,2 K) is 1,59 (7) g / cm³
Density of solid ozone at −195,7 ° С (77,4 К) is equal to 1,73 (2) g / cm³
Melting point −197,2 (2) ° С (75,9 К)
For a long time, engineers suffered with him, trying to use as a high-energy and at the same time environmentally friendly oxidant in rocket technology.
The total chemical energy released by the combustion reaction involving ozone is greater than for simple oxygen, by about one quarter (719 kcal / kg). More will be, respectively, and Jud. Liquid ozone has a higher density than liquid oxygen (1,35 versus 1,14 g / cm³, respectively), and its boiling temperature is higher (−112 ° C and −183 ° C, respectively).
So far, an insurmountable obstacle is the chemical instability and explosion hazard of liquid ozone, decomposing it into O and O2, in which a detonation wave moving at a speed of about 2 km / s and destructive detonation pressure develops more than 3 · 107 dyn / cm2 (3 MPa) making the use of liquid ozone is impossible with the current level of technology, with the exception of the use of stable oxygen-ozone mixtures (up to 24% ozone). The advantage of this mixture is also a greater specific impulse for hydrogen engines, compared with ozone-hydrogen. To date, such high-performance engines as the RD-170, RD-180, RD-191, as well as booster vacuum engines have reached IU on parameters close to the limiting values, and to increase the UI, there is only one possibility associated with the transition to new fuels .
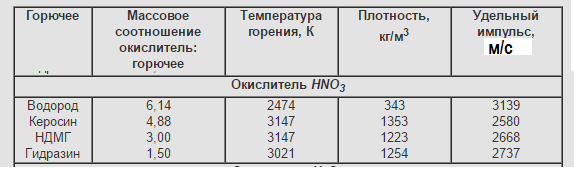
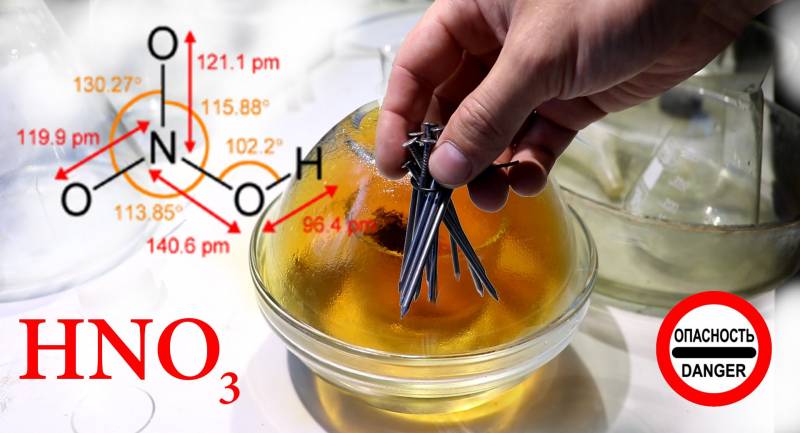
Nitric acid-HNO3
The molar mass of 63.012 g / mol (no matter what I use molar mass or molecular weight is not changing the essence)
Density = 1,513 g / cm³
T. melt = - 41,59 ° C, T. kip. = 82,6 ° C
HNO3 has a high density, low cost, is produced in large quantities, is quite stable, including at high temperatures, fire and explosion proof. Its main advantage over liquid oxygen in high boiling point, and, consequently, in the possibility of being stored indefinitely without any thermal insulation. HNO nitric acid molecule3 - almost perfect oxidizer. It contains as a “ballast” a nitrogen atom and a “half” of a water molecule, and two and a half oxygen atoms can be used to oxidize fuel. But it was not there! Nitric acid is so aggressive that it continuously reacts with itself — hydrogen atoms split off from one acid molecule and join adjacent ones, forming fragile, but extremely chemically active aggregates. Even the most resistant grades of stainless steel are slowly destroyed by concentrated nitric acid (as a result, a thick greenish jelly, a mixture of metal salts, was formed at the bottom of the tank). To reduce the corrosivity of steel, various substances are added to nitric acid; in total, 0,5% hydrofluoric (hydrofluoric) acid reduces the corrosion rate of stainless steel by a factor of ten.
For almost 20 years we have been looking for a suitable container for nitric acid. It is very difficult to select construction materials for tanks, pipes, and LRE combustion chambers.
An option of an oxidizing agent that was chosen in the USA, with 14% nitrogen dioxide. And our missilemen acted differently. It was necessary to catch up with the United States at all costs, so the oxidizers of the Soviet brands - AK-20 and AK-27 - contained 20 and 27% tetroxide.
Interesting fact: In the first Soviet rocket fighter BI-1, nitric acid and kerosene were used for flights.

Tanks and pipes had to be made from monel metal: an alloy of nickel and copper, it became a very popular construction material among the rocket men. Soviet rubles were almost 95% made of this alloy.
Disadvantages: tolerant "disgusting." Corrosive active. The specific impulse is not high enough. Currently in its pure form is almost never used.
Nitrogen tetroxide-AT (N2O4)
Density = 1,443 g / cm³
"Took the baton" from nitric acid in military engines. Possesses saomovosplamenemost with hydrazine, UDMH. Low-boiling component, but can be stored for a long time when taking special measures.
Disadvantages: the same crap as HNO3but with its quirks. May decompose to nitric oxide. Toxic. Low specific impulse. Often used and used oxidant AK-NN. It is a mixture of nitric acid and nitric tetroxide, sometimes referred to as "red fuming nitric acid." Numbers indicate percentage N2O4.
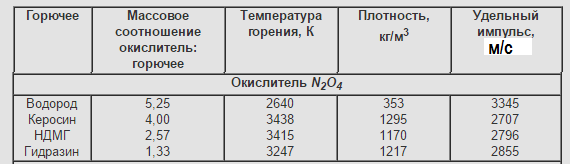
Basically, these oxidizing agents are used in the LRE for military purposes and the LRE spacecraft due to their properties: long-duration and self-igniting. Characteristic combustible for AT is UDMH and hydrazine.
Fluorine-F2
Molar mass F2, 37,997 g / mol
Melting point = 53,53 K (−219,70 ° C)
Boiling point = 85,03 K (−188,12 ° C)
Density (for the liquid phase), ρ = 1,5127 g / cm³
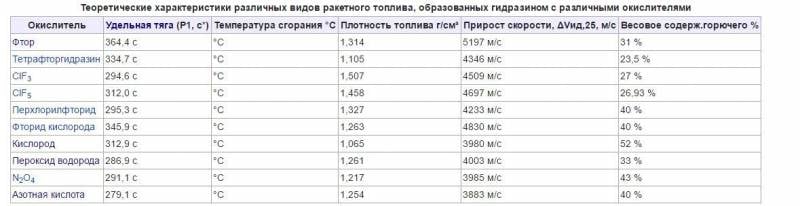
Fluorine chemistry began to develop since 1930-ies, especially quickly - during the years of 2-th world war 1939-45 and after it in connection with the needs of the nuclear industry and rocket technology. The name "Fluorine" (from the Greek. Phthoros - destruction, death), proposed by A. Ampere in the 1810 year, is used only in Russian; In many countries, adopted the name "Fluor". This is an excellent oxidizing agent in terms of chemistry. Oxidizes and oxygen, and water, and in general almost everything. Calculations show that the maximum theoretical Iud can be obtained on a pair of F2-Be (beryllium) -6000 order m / s!
Super? Bummer, not "super" ...
The enemy does not want such an oxidizer.Extremely corrosive, toxic, prone to explosions when in contact with oxidizing materials. Cryogenic. Any product of combustion also has almost the same "sins": terribly corrosive and toxic.
Safety Instructions Fluorine is toxic, its maximum permissible concentration in air is about 2 · 10-4 mg / l, and the maximum permissible concentration at exposure of no more than 1 h is 1,5 · 10-3mg / l.
LRE 8Д21 use of a pair of fluorine + ammonia gave a specific impulse at the level of 4000 m / s.
For a pair of F2+H2 it turns out Iud = 4020 m / s!
The trouble: HF-hydrogen fluoride on the "exhaust".
Starting position after the launch of such an "energetic engine"?
A pool of liquid metals and other chemical and organic objects dissolved in hydrofluoric acid!
Н2+ 2F = 2HF, at room temperature, exists as H dimer2F2.
It is mixed with water in any respect with the formation of hydrofluoric (hydrofluoric) acid. And its use in the LRE KA is not realistic because of the murderous complexity of storage and the destructive action of combustion products.
All the same applies to the remaining liquid halogens, for example, to chlorine.

Hydrogen fluoride LRE 25 t for equipping both stages of the rocket booster AKC Spiral supposed to be developed in OKB-456 V.P. Glushko on the basis of spent LRE 10 t on fluoroammic (F2+ NH3) fuel.
Hydrogen peroxide-H2O2.
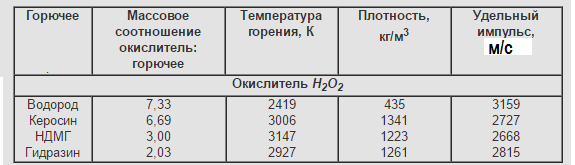
It is mentioned by me above in single-component fuels.
Walter HWK 109-507: advantages in the simplicity of the LRE design. A vivid example of such a fuel is hydrogen peroxide.
Hydrogen peroxide for luxurious hair "natural" blondes and 14 more secrets of its use.

Alles: the list of more or less real oxidants is over. Focusing on HClО4. As independent oxidants based on perchloric acid, they are of interest only: monohydrate (H2O + ClO4) - solid crystalline substance and dihydrate (2NO + HClO4) - tight viscous fluid. Perchloric acid (which is unpromising in itself due to Iod), is of interest as an additive to oxidizing agents, which guarantees the reliability of self-ignition of fuel.
Oxidizers can be classified as follows:

The final (more commonly used) list of oxidizers in conjunction with the real flammable ones:
Note: if you want to translate one variant of specific impulse into another, you can use a simple formula: 1 m / s = 9,81 with.
In contrast, they are combustible here. "heaps".
Combustible
The main characteristics of two-component LRT at pc / pa = 7 / 0,1 MPa
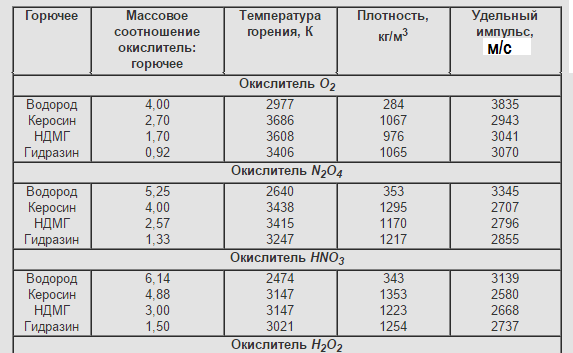
According to the physico-chemical composition, they can be divided into several groups:
Low molecular weight hydrocarbons.
Simple substances: atomic and molecular.
For this topic, for the moment, only hydrogen (Hydrogenium) is of practical interest.
Na, Mg, Al, Bi, He, Ar, N2Br2Si, Cl2, The2 and others. I will not consider in this article.
Hydrazine fuels ("stinkers").
Wake up to Sony - we got to alcohol (С2Н5ОН).
The search for the optimal fuel began with the development of LRE enthusiasts. The first widely used fuel was ethanol)used first
Soviet missiles Р-1, Р-2, Р-5 ("legacy" ФАУ-2) and on Vergeltungswaffe-2 itself.
Rather, the solution 75% ethyl alcohol (ethanol, ethyl alcohol, methyl carbinol, wine alcohol or alcohol, often simply colloquially "alcohol") is a monohydric alcohol with formula C2H5OH (empirical formula C2H6O), another option: CH3-CH2-OH
This fuel two serious drawbacks, which obviously did not suit the military: low energy indicators and low resistance of personnel to the "poisoning" of such fuel.
Supporters of a healthy lifestyle (spirits) tried to solve the second problem with the help of furfuryl alcohol. It is poisonous, mobile, transparent, sometimes yellowish (to dark brown), with time a liquid reddening in air. BARNES!
Chem. formula: C4H3AND2OH, Rat. formula: C5H6O2. Disgusting swill. To drink is not suitable.
Group of hydrocarbons.
Kerosene
Combustible mixture of liquid hydrocarbons (from C8 to C15) with a boiling point in the range 150 — 250 ° C, clear, colorless (or slightly yellowish), slightly oily to the touch
Density - from 0,78 to 0,85 g / cm³ (at temperature 20 ° С);
viscosity - from 1,2 - 4,5 mm² / s (at temperature 20 ° С);
flash point from 28 ° С to 72 ° С;
calorific value - 43 MJ / kg.
My opinion: it’s pointless to write about the exact molar mass
Kerosene is a mixture of various hydrocarbons, so there are terrible fractions (in the chemical formula) and the "smeared" boiling point. Convenient high boiling fuel. Used for a long time and successfully worldwide in engines and in aviation. It is on it that the Unions still fly. Low toxicity (strongly do not recommend drinking), stable. Nevertheless, kerosene is dangerous and unhealthy (ingestion).
But there are people who treat them nothing! Ministry of Health strongly opposed!
Soldier tales: well helps to get rid of nasty Pthirus pubis.
However, it also requires caution when handling: passenger aircraft crash video
Significant advantages: relatively inexpensive, mastered in production. A kerosene-oxygen pair is ideal for the first stage. Its specific impulse on the ground is 3283 m / s, the empty 3475 m / s. Disadvantages. Relatively low density.
American Rocket Kerosene Rocket Propellant-1 or Refined Petroleum-1
About cheap was before.
To increase the density, the leaders of space exploration were developed by Sintine (USSR) and RJ-5 (USA).
Sintine synthesis.
Kerosene has a tendency to deposit tar in the highways and the cooling path, which has a negative effect on cooling. At this his bad property pedal Mukhin, Velor @Co.
Kerosene engines are most mastered in the USSR.
A masterpiece of human reason and engineering our "pearl" RD-170 / 171:
Now the term has become a more correct name for combustible on the basis of kerosene HCG- "hydrocarbon fuel", because from kerosene, which was burned in the safe kerosene lamps of I. Lukasevich and J. Zeh, the UVG used "went away" very long away.
As an example:naphthyl.
In fact, "Roskosmos" dezu gives out:
Low molecular weight hydrocarbons
Methane-CH4
Density of gas (0 ° C) 0,7168 kg / m³;
liquid (−164,6 ° C) 415 kg / m³
T. melt = - 182,49 ° C
T. Kip. = - 161,58 ° C
Everyone is now considered as a promising and cheap fuel, as an alternative to kerosene and hydrogen.
Chief Designer NPO Energomash Vladimir Chvanov:
Inexpensive, common, stable, low toxic. Compared to hydrogen, it has a higher boiling point, and the specific impulse paired with oxygen is higher than that of kerosene: about 3250-3300 m / s on the ground. Good cooler.
Disadvantages. Low density (twice lower than that of kerosene). In some modes of combustion, it can decompose with carbon in the solid phase, which can lead to a drop in the pulse due to the two-phase flow and a sharp deterioration in the cooling mode in the chamber due to the deposition of soot on the walls of the CS. Recently, active NOR and R & D in the field of its application (along with propane and natural gas), even in the direction of modification, are already existing. LRE (in particular, such work was carried out on RD-0120).

Or "Kinder Surpeis", as an example: Space X American Raptor engine:
These fuels include propane and natural gas. Their main characteristics, like combustible, are close (with the exception of greater density and higher boiling point) to HCG. And there are the same problems with their use.
Apart among combustible positioned Hydrogen-H2 (Liquid: lh2).
Density (when n. U.) = 0,0000899 (at 273 K (0 ° C)) g / cm³
Melting point = 14,01K (-259,14 ° C);
Boiling point = 20,28K (-252,87 ° C);
Use of a LOX-LH pair2 proposed by Tsiolkovsky, but implemented by others:
In terms of thermodynamics H2 an ideal working body for both the LRE itself and the TNA turbine. Excellent cooler, with both in the liquid and in the gaseous state. The latter fact makes it possible not to be particularly afraid of the boiling of hydrogen in the cooling path and to use the gasified hydrogen in this way to drive the THA.
This scheme is implemented in the Aerojet Rocketdyne RL-10-just smart (from an engineering point of view) engine:
Our counterpart (even betterbecause younger): RD-0146 (D, DM) is a gas-free liquid-propellant rocket engine developed by the Design Bureau of Chemical Automation in Voronezh.

Especially effective with a nozzle nozzle from the material "Grauris". But not yet fly
This TC provides a high specific impulse-paired with oxygen 3835 m / s.
Of the actually used is the highest rate. These factors cause a keen interest in this fuel. Environmentally friendly, at the "outlet" in contact with O2: water (water vapor). Distributed, almost unlimited reserves. Mastered in production. Non-toxic. However, there are a lot of fly in the ointment in this barrel of honey.
1. Extremely low density. Everyone saw the huge hydrogen tanks of the PH Energia and the Space Shuttle Space Shuttle. Due to its low density, it is applicable (as a rule) in the upper stages of the LV.
In addition, low density poses a difficult task for pumps: multi-stage hydrogen pumps to ensure the required mass flow and not to cavitate.
For the same reason, you have to put a so-called. booster fuel pumping units (BNAG) immediately behind the intake device in the tanks, in order to facilitate the life of the main TNA.

Also, hydrogen pumps for optimal conditions require a significantly higher rotational speed of the TNA.
2. Low temperature. Cryogenic fuel. Before refueling, it is necessary to carry out many hours of cooling down (and / or overcooling) of tanks and the entire path. Bucky PH "Falocn 9FT" - inside view:
More about "surprises":
"MATHEMATICAL MODELING OF HEAT AND MASS-EXCHANGE PROCESSES IN HYDROGEN SYSTEMS" H0P VA GordeevV.P. Firsov, A.P. Gnevashev, E.I. Postoyuk
FSUE "GKNPTs them. Mv Khrunicheva, Salyut; "Moscow Aviation Institute (State Technical University)
The low boiling point makes it difficult to pump into tanks and store this fuel in tanks and storages.
3. Liquid hydrogen has some gas properties:
Hydrogen can be in ortho and para states. Orthohydrogen (o-H2) has a parallel (of the same sign) orientation of nuclear spins. Para-hydrogen (p-H2) -anti-parallel.
At normal and high temperatures H2 (normal hydrogen, n-Н2) is a mixture of 75% ortho- and 25% para-modifications, which can mutually turn into each other (ortho-para-transformation). When turning oh2 in pn2 heat is released (1418 j / mol).
This all imposes additional difficulties in the design of highways, LRE, THA, cyclograms of work, and especially pumps.
4. Hydrogen gas, faster than other gases, spreads through space, passes through small pores, and at high temperatures penetrates steel and other materials relatively easily. H2g has a high thermal conductivity, equal when 273,15 K and 1013 hPa 0,1717 W / (m * K) (7,3 with respect to air).
Hydrogen in the normal state at low temperatures is inactive, without heating it reacts only with F2 and in the light with cl2. Hydrogen interacts with non-metals more actively than with metals. It reacts with oxygen almost irreversibly, forming water with release of 285,75 MJ / mol of heat;
5. With alkaline and alkaline-earth metals, elements of group III, IV, V, and VI of the periodic system, as well as with intermetallic compounds, hydrogen forms hydrides. Hydrogen reduces the oxides and halides of many metals to metals, unsaturated hydrocarbons to saturated ones (see Hydrogenation).
Hydrogen gives up its electron very easily. In solution, it detaches in the form of a proton from many compounds, causing their acidic properties. In aqueous solutions, H + forms a hydroxonium ion H with a water molecule.3A. Being part of the molecules of various compounds, hydrogen tends to form a hydrogen bond with many electronegative elements (F, O, N, C, B, Cl, S, P).
6. Fire hazard and explosiveness. You can not rassusolivat: everyone knows an explosive mixture.
A mixture of hydrogen with air explodes from the slightest spark in any concentration - from 5 to 95 percent.
So there is hydrogen and gut (even Excellent), and at the same time "headache" (even severe headache).
The first law of dialectics: "Unity and the struggle of opposites" /Georg Wilhelm Friedrich Hegel/
Impressive Space Shuttle Main Engine (SSME)?
Now estimate its cost!
Probably, after seeing this and considering the costs (the cost of putting the 1 kg into PN orbit, the legislators and those who steer the US budget and NASA in particular ... decided "well, it's in FIG."
And I understand them - on the Soyuz RN it is both cheaper and safer, and using the RD-180 / 181 removes many of the problems of American RN and significantly saves the taxpayers ’money in the richest country in the world.
The most mastered hydrogen engines in the United States.
Now we are positioning on the 3-4 place in the “Hydrogen Club” (after Europe, Japan and China / India).
Separately mention solid and metallic hydrogen.

Solid hydrogen crystallizes in a hexagonal lattice (a = = 0,378 nm, c = 0,6167 nm), in the nodes of which H molecules are located2interconnected weak intermolecular forces; density 86,67 kg / m³; C ° 4,618 J / (mol * K) at 13 K; dielectric. At pressures above 10000 MPa, a phase transition is expected with the formation of a structure built of atoms and possessing metallic properties. Theoretically predicted the possibility of superconductivity "metallic hydrogen".
Melting point −259,2 ° C (14,16 K).
Density 0,08667 g / cm³ (at −262 ° C).
White snow-like mass, crystals of hexagonal syngony.
The Scottish chemist J. Dewar in the 1899 for the first time received hydrogen in a solid state. For this, he used a regenerative cooling machine based on the effect Joule-Thomson.
Trouble with him. He is constantly lost: "Scientists have lost the world's only sample of metallic hydrogen". It is understandable: a cube was obtained from molecules: 6х6х6. Just "giant" volumes - right now, "refill" the rocket. For some reason it reminded me "Nanotank of Chubais". This nano miracle can not find already 7 years or more.
Anamason, antimatter, metastable helium while I leave behind the scenes.
...
Hydrazine Fuels ("Stinkers")
Hydrazine-N2H4
A condition at NU. - colorless liquid
Molar mass = 32.05 g / mol
Density = 1.01 g / cm³
Very common fuel.
It is stored for a long time, and it is “loved” for it. It is widely used in remote control of spacecraft and ICBM / SLBM, where long-duration is of critical importance.
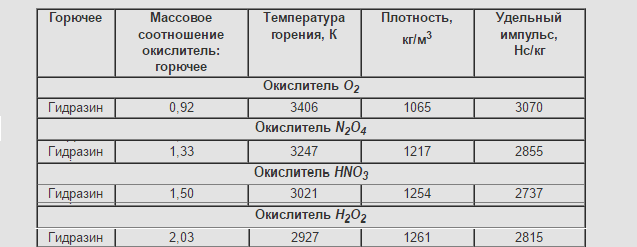
Who was embarrassed by Iud in the dimension of H * s / kg I answer: this designation is "loved" by the military.
Newton is a derived unit, based on Newton's second law it is defined as the force that changes the speed of a body with a mass of 1 kg per 1 m / s in the direction of the force in 1 seconds. Thus, 1 H = 1 kg · m / s2.
Accordingly: 1 N * s / kg = 1 kg · m / s2* s / kg = m / s.
Mastered in production.

Disadvantages: toxic, smelly.
Hydrazine vapor explodes during adiabatic compression. It is prone to decomposition, which, however, allows it to be used as a monofuel for small thrust (LPDMT) thrusters. Due to the development of production is more common in the United States.
Asymmetric dimethyl hydrazine (UDMH) -H2NN (CH3)2
A condition at NU - liquid
Molar mass = 60,1 g / mol
Density = 0,79 ± 0,01 g / cm³
Widely used on military engines as a result of its durability. With the development of technology ampulyatsii - almost all problems have disappeared (except for disposal and accidents allowances).
It has a higher impulse compared to hydrazine.
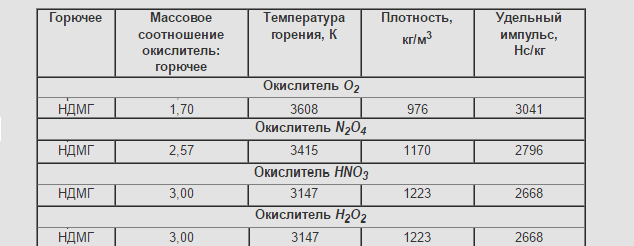
Density and specific impulse with main oxidants below kerosene with the same oxidizers. Self-ignite with nitric oxidants. Mastered in production in the USSR.
Favorite fuel V.P.Glushko. Not the favorite fuel of my OZK and the surrounding wildlife.

I can write a whole article about its nasty properties (based on the operation of the C-200 air defense system).
It is used, as a rule, with nitric oxidants on LRE MBR, SLBMs, KA, and on our Proton- *.

Disadvantages: extremely toxic. The same "skunk", like the rest of the "skunk". An order of magnitude more expensive than kerosene.

Hydrazine is extremely poisonous
To increase the density is often used in a mixture with hydrazine, the so-called. aerosin-50, where 50 is the percentage of UDMH. More common in the USSR.
And in the jet engine of a French fighter-bomber Dassault Mirage III (I recommend a good video) UDMH is used as an activating additive to traditional fuels.
About hydrazine fuels.
The specific thrust is equal to the ratio of thrust to weight fuel consumption; in this case, it is measured in seconds (s = N · s / N = kgf · s / kgf). To convert the weight specific gravity to the mass, it must be multiplied by the acceleration of gravity (approximately equal to 9,81 m / s²)
Behind the scenes left:
Aniline, methyl, dimethyl and trimethylamines and CH3NHNH2-Methylhydrazine (aka monomethylhydrazine or heptyl), etc.
In professional jargon, these fuels are called "stinky" or "stinky."
It can be said with a high degree of confidence that if there are “smelly” engines on the LV, then "before marriage" she was a combat missile (ICBM, SLBM or anti-aircraft missiles - which is already rare). Chemistry in the service and the army and the citizen.

The only exception is, perhaps, the Ariane PH - the creation of a cooperative: Aérospatiale, Matra Marconi Space, Alenia, Spazio, DASA, etc. It was passed away like a similar fate in the “girlhood”.
The military almost all switched to solid-propellant rocket motors, as more convenient to operate. The niche for smelly fuels in the space program has narrowed down to use in the spacecraft remote control, where long-term storage is required without any special material or energy costs.
Perhaps a brief overview can be expressed graphically:
Rocket men are actively working with methane. There are no particular operational difficulties: it allows for a good pressure increase in the chamber (up to 40 MPa) and get good performance.
(РД0110МД, РД0162. Methane projects. Perspective reusable launch vehicles) and other natural gases (LNG).
On other directions to improve the characteristics of the LRE (combustible metallization, the use of He2, acetam and other) I will write later. If there is interest.
Detonation burning is an opportunity for a long-awaited jump to Mars.
Afterword:
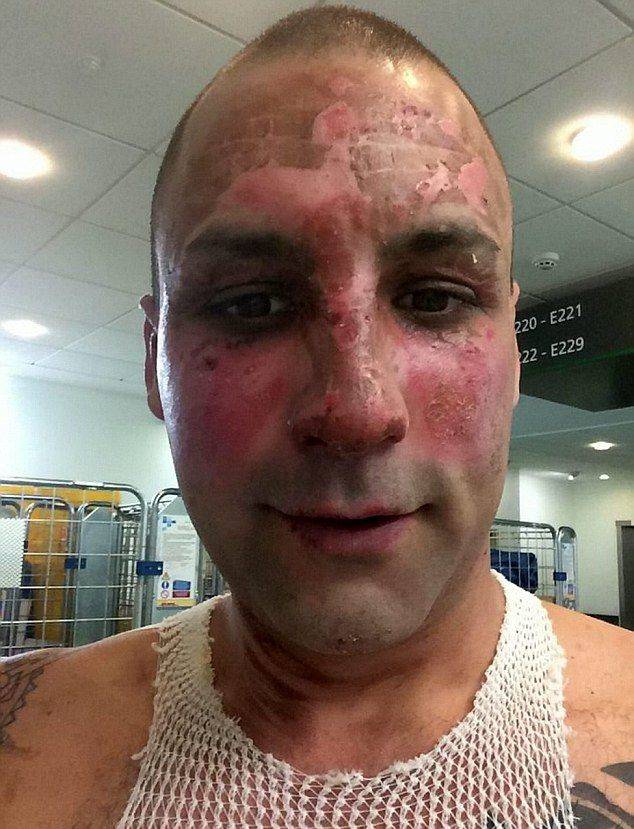
in general, all missile launchers (except for NTK), as well as an attempt to make them at home, are very dangerous. I suggest to carefully read:26-year-old Chris Monger, father of two children, decided to prepare rocket fuel at home, following instructions, spied on YouTube. The mixture, which he cooked on the stove in a saucepan, exploded as expected. As a result, the man received a huge number of burns and spent five days in the hospital.
All household (garage) manipulations with such chemical components are extremely dangerous, and sometimes illegal. It is BETTER to not approach the places of their spill without OZK and gas mask:
As with spilled mercury: to call the Ministry of Emergency Situations, they will quickly arrive and they will pick everything up professionally.
Thank you all who could endure all this to the end.
Primary Sources:
Kachur P. I., Glushko A.V. "Valentin Glushko. Designer of rocket engines and space systems", 2008.
G.G. Gahun "Design and design of liquid rocket engines", Moscow, "Mechanical Engineering, 1989.
The possibility of increasing the specific impulse of a liquid rocket engine
when helium SA is added to the combustion chamber. Orlin MGTU them. N.E. Bauman, Moscow
M.S.Shehter. "Fuel and working bodies of rocket engines", Mechanical Engineering "1976
Zavistovsky D. I. "Conversations about rocket engines".
Phillip Terekhov @lozga (www.geektimes.ru).
"Types of fuel and their characteristics. Fuel is combustible substances used to produce heat. Fuel composition The fuel part is carbon C-hydrogen H-sulfur." - Oksana Kaseeva
Fakas SS "Fundamentals of the LPRE. Working body"
Photo and video materials from sites are used:
The television studio of Roskosmos
http://technomag.bmstu.ru
www.abm-website-assets.s3.amazonaws.com
www.free-inform.ru
www.rusarchives.ru
www.epizodsspace.airbase.ru
www.polkovnik2000.narod.ru
www.avia-simply.ru
www.arms-expo.ru
www.npoenergomash.ru
www.buran.ru
www.fsmedia.imgix.net
www.wikimedia.org
www.youtu.be
www.cdn.tvc.ru
www.commi.narod.ru
www.dezinfo.net
www.nasa.gov
www.novosti-n.org
www.prirodasibiri.ru
www.radikal.ru
www.spacenews.com
www.esa.int
www.bse.sci-lib.com
www.kosmos-x.net.ru
www.rocketpolk44.narod.ru
www.criotehnika.ru
www.transavtocisterna.rf
www.chistoprudov.livejournal.com/104041.html
www.cryogenmash.ru
www.eldeprocess.ru
www.chemistry-chemists.com
www.rusvesna.su
www.arms-expo.ru
www.armedman.ru
www.transavtocisterna.rf
www.ec.europa.eu
www.mil.ru
www.kbkha.ru
www.naukarus.com



















































Information